
Chemistry, 12.02.2020 04:39 winterblanco
1. Use VSEPR theory to decide which one of the following ions and molecules is likely to be planar. (The central atom is always first in the formula.) A. BrF3 B. H3O C. PCl3 D. SO42- E. SF4

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:00, Ezekielcassese
What is the percentage composition of carbon in the compound ch4
Answers: 1

Chemistry, 22.06.2019 21:50, namoralessimon03
28. which is not a reason that water is used to store spent fuel rods from nuclear power plants? water increases the speed of the chain reaction in the fuel rods. water protects nuclear power plant workers from the high temperature and radiation of the fuel rods. water acts as a radiation shield to reduce the radiation levels. water cools the spent rods. salts action
Answers: 1

Chemistry, 22.06.2019 22:30, safiyabrowne7594
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1
You know the right answer?
1. Use VSEPR theory to decide which one of the following ions and molecules is likely to be planar....
Questions in other subjects:

English, 26.09.2019 02:10



Arts, 26.09.2019 02:10


Mathematics, 26.09.2019 02:10

History, 26.09.2019 02:10

History, 26.09.2019 02:10

Social Studies, 26.09.2019 02:10

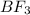
![\text{Number of electron pair}=\frac{1}{2}[V+N-C+A]](/tpl/images/0508/1323/9e987.png)
![\text{Number of electrons}=\frac{1}{2}\times [3+3]=3](/tpl/images/0508/1323/ce6da.png)
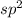 and the electronic geometry of the molecule will be trigonal planar.
and the electronic geometry of the molecule will be trigonal planar.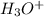
![\text{Number of electrons}=\frac{1}{2}\times [6+3-1]=4](/tpl/images/0508/1323/9514c.png)
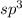 and the electronic geometry of the molecule will be tetrahedral.
and the electronic geometry of the molecule will be tetrahedral.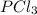
![\text{Number of electrons}=\frac{1}{2}\times [5+3]=4](/tpl/images/0508/1323/5f4d9.png)
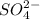
![\text{Number of electrons}=\frac{1}{2}\times [6+2]=4](/tpl/images/0508/1323/2c46b.png)
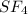
![\text{Number of electrons}=\frac{1}{2}\times [6+4]=5](/tpl/images/0508/1323/f1dca.png)
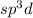 and the electronic geometry of the molecule will be tetrahedral.
and the electronic geometry of the molecule will be tetrahedral.


