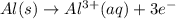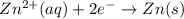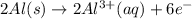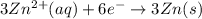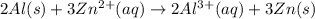This is an incomplete question, here is a complete question.
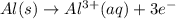
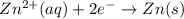
The half-reactions for the oxidation-reduction reaction between Al(s) and Zn²⁺(aq) are represented above. Based on the half-reactions, what is the coefficient for Al(s) if the equation for the oxidation-reduction reaction is balanced with the smallest whole-number coefficients?
Answer : The coefficient for Al(s) is, 2
Explanation :
Redox reaction or Oxidation-reduction reaction : It is defined as the reaction in which the oxidation and reduction reaction takes place simultaneously.
Oxidation reaction : It is defined as the reaction in which a substance looses its electrons. In this, oxidation state of an element increases. Or we can say that in oxidation, the loss of electrons takes place.
Reduction reaction : It is defined as the reaction in which a substance gains electrons. In this, oxidation state of an element decreases. Or we can say that in reduction, the gain of electrons takes place.
The given oxidation-reduction half reaction are :
Oxidation : 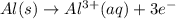
Reduction : 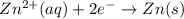
Now balance the charge.
In order to balance the electrons, we multiply the oxidation reaction by 2 and reduction reaction by 3 and then added both equation, we get the balanced redox reaction.
Oxidation : 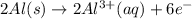
Reduction : 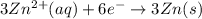
The balanced chemical equation will be,
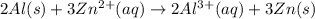
Thus, the coefficient for Al(s) is, 2