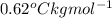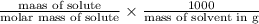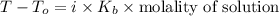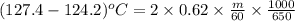A certain liquid has a normal boiling point of and a boiling point elevation constant . A solution is prepared by dissolving some sodium chloride () in of . This solution boils at . Calculate the mass of that was dissolved. Round your answer to significant digits.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, nana54muller
Part 1: include important facts found through your research. part 2: include your visual display. include your summary of “the chemistry of water” from the national science foundation website. include your experiment. part 3: include responses to the reflection questions.
Answers: 1

Chemistry, 22.06.2019 19:10, krisandlance
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1

Chemistry, 23.06.2019 12:30, asseatingbandit
You have 125 g of a certain seasoning and are told that it contains 70.0 g of salt. what is the peroentage of salt by mass in this seasoning?
Answers: 2

Chemistry, 23.06.2019 14:00, wendyyy1214
If you fill your car tire to a pressure of 32 psi (pounds per square inch) on a hot summer day when the temperature is 35°c (95°f), what is the pressure (in psi) on a cold winter day when the temperature is -15°c (5°f)? assume no gas leaks out between measurements and the volume of the tire does not change.
Answers: 1
You know the right answer?
A certain liquid has a normal boiling point of and a boiling point elevation constant . A solution i...
Questions in other subjects:

Social Studies, 05.08.2019 16:10

Social Studies, 05.08.2019 16:10

Computers and Technology, 05.08.2019 16:10

English, 05.08.2019 16:10

Physics, 05.08.2019 16:10



English, 05.08.2019 16:10

Mathematics, 05.08.2019 16:10

Business, 05.08.2019 16:10

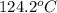 and a boiling point elevation constant
and a boiling point elevation constant 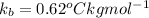 . A solution is prepared by dissolving some sodium chloride (NaCl) in 6.50 g of X. This solution boils at
. A solution is prepared by dissolving some sodium chloride (NaCl) in 6.50 g of X. This solution boils at 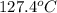 . Calculate the mass of NaCl that was dissolved. Round your answer to significant digits.
. Calculate the mass of NaCl that was dissolved. Round your answer to significant digits.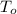 = boiling point of the pure solvent =
= boiling point of the pure solvent = 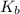 = elevation of boiling constant =
= elevation of boiling constant =