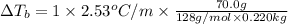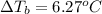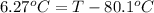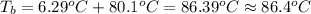Calculate the boiling point of a solution prepared by dissolving 70.0 g of naphthalene, c10h8 (a nonvolatile nonelectrolyte), in 220.0 g of benzene, c6h6. the kb for benzene = 2.53oc/m. the boiling point of pure benzene is 80.1oc.. ans: 86.4 degrees celsius. i did this so far. 1)70g c10h8(1mol c10h8/128gc10h8)= .546mol c10h8. benzene)=2.482m. 3) (2.482)(2.53 c/m)=6.289. i'm not sure if i am starting this off right, can anyone me get the correct answer? ans ty!

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:30, darkghostmist
What type of reaction fuels the processes seen here?
Answers: 2


Chemistry, 23.06.2019 01:30, joyelewis58
Will a solution form when the solvent and solute are both nonpolar? a. not likely b. never c. most likely
Answers: 1

Chemistry, 23.06.2019 03:30, Ramann03
27 drag each label to the correct location on the image. a particular exosolar system has five planets in total: a, b, c, d, and e. the table lists the orbital periods of these planets in days. planet orbital period (days) a 600 b 80 c 1,000 d 500 e 100 move each planet to its orbit in the system.
Answers: 3
You know the right answer?
Calculate the boiling point of a solution prepared by dissolving 70.0 g of naphthalene, c10h8 (a non...
Questions in other subjects:


Mathematics, 17.10.2019 20:40

Mathematics, 17.10.2019 20:40

Mathematics, 17.10.2019 20:40

English, 17.10.2019 20:40



Mathematics, 17.10.2019 20:40

Mathematics, 17.10.2019 20:40

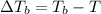
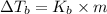
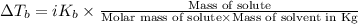
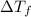 =Elevation in boiling point
=Elevation in boiling point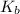 = Boiling point constant of solvent = 2.53 °C/m(benzene)
= Boiling point constant of solvent = 2.53 °C/m(benzene)