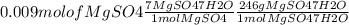Chemistry, 29.01.2020 04:50 taylabrown2013
On the first day of your new job as a chemist, you are given a bottle of magnesium sulfate and asked to make 30 ml of 0.3 m mgso4. the formula on the bottle is mgso4∗7h2o (also known as epsom salt). calculate the amount of salt you need (in milligrams).

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, actheorian8142
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3


Chemistry, 23.06.2019 02:30, roseemariehunter12
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3

Chemistry, 23.06.2019 03:00, draveon6925
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2
You know the right answer?
On the first day of your new job as a chemist, you are given a bottle of magnesium sulfate and asked...
Questions in other subjects:

Biology, 24.06.2019 00:00

Mathematics, 24.06.2019 00:00

History, 24.06.2019 00:00

History, 24.06.2019 00:00



Mathematics, 24.06.2019 00:00

History, 24.06.2019 00:00

Biology, 24.06.2019 00:00