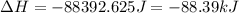Chemistry, 28.01.2020 00:31 kyzerlinda
Based on the thermodynamic properties provided for water, determine the amount of energy released for 155.0 g of water to go from 39.0 °c to -36.5°c. property melting point boiling point ahfus ahvap cp (s) value 0.0 100.0 6.01 40.67 37.1 75.3 33.6 units oc kj/mol kj/mol j/mol. oc j/mol c mol oc cp (g)

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 08:40, kellymcdow5135
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3
You know the right answer?
Based on the thermodynamic properties provided for water, determine the amount of energy released fo...
Questions in other subjects:

Business, 14.11.2020 19:20

Mathematics, 14.11.2020 19:20

Mathematics, 14.11.2020 19:20




Social Studies, 14.11.2020 19:20

Social Studies, 14.11.2020 19:20

Geography, 14.11.2020 19:20

Advanced Placement (AP), 14.11.2020 19:20

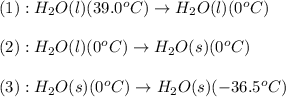
![\Delta H=[m\times c_{p,l}\times (T_{final}-T_{initial})]+m\times (-\Delta H_{fusion})+[m\times c_{p,l}\times (T_{final}-T_{initial})]](/tpl/images/0474/1272/1181a.png)
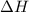 = heat available for the reaction = ?
= heat available for the reaction = ?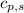 = specific heat of solid water =
= specific heat of solid water = 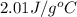
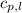 = specific heat of liquid water =
= specific heat of liquid water = 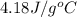
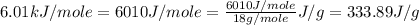
![\Delta H=[155.0g\times 4.18J/g^oC\times (0-(39.0))^oC]+155.0g\times -333.89J/g+[155.0g\times 2.01J/g^oC\times (-36.5-0)^oC]](/tpl/images/0474/1272/a7829.png)