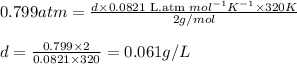Chemistry, 27.01.2020 22:31 SkinnestXOXO
Asample of hydrogen gas has a density of g/l at a pressure of 0.799 atm and a temperature of 47 °c. assume ideal behavior.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, annsmith66
What is the result of multiplying (2.5 × 1010) × (2.0 × 10-7)? a. 5.0 × 103 b. 5.0 × 10-3 c. 5.0 × 1017 d. 5.0 × 10-17
Answers: 1


Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 22.06.2019 23:00, DESI111609
What is the average rate of the reaction between 10 and 20 s?
Answers: 1
You know the right answer?
Asample of hydrogen gas has a density of g/l at a pressure of 0.799 atm and a temperature of 47 °c....
Questions in other subjects:

Social Studies, 16.10.2020 19:01

Physics, 16.10.2020 19:01


Biology, 16.10.2020 19:01

English, 16.10.2020 19:01

History, 16.10.2020 19:01


Mathematics, 16.10.2020 19:01



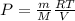
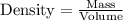
 ......(1)
......(1)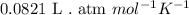
![47^oC=[47+273]K=320K](/tpl/images/0473/6736/7e1e2.png)