
Chemistry, 25.01.2020 06:31 jaleewoodyard1
Apharmacist–herbalist mixed 100 g lots o st. john’s wort containing the ollowing percentages o the active component hypericin: 0.3%, 0.7%, and 0.25%. calculate the percent strength o hypericin in the mixture.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:00, nothingworksoutforme
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 12:30, pup88
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2
You know the right answer?
Apharmacist–herbalist mixed 100 g lots o st. john’s wort containing the ollowing percentages o the a...
Questions in other subjects:



Biology, 11.03.2021 18:20







English, 11.03.2021 18:20

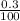 × 100
× 100 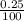 × 100
× 100 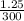 × 100
× 100 

