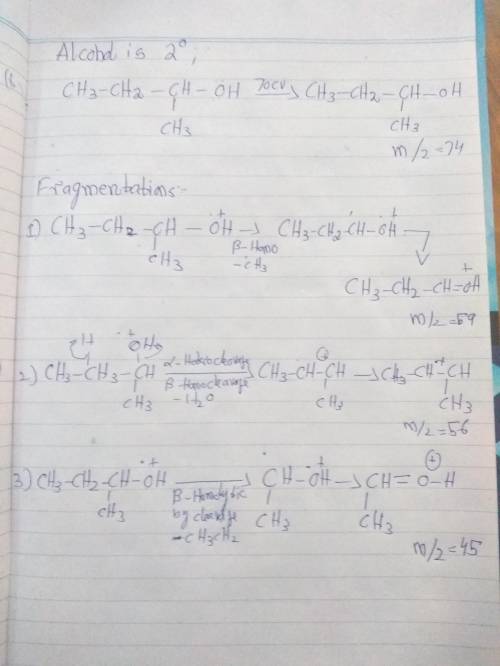Chemistry, 25.01.2020 03:31 maxraph108
Draw a structure for an alcohol that exhibits a molecular ion at m+ = 74 and that produces fragments at m/z = 59, m/z = 56 and m/z = 45.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:00, princessroyal
This graph gives information on changes in sea ice extent in the arctic ocean over a 30-year span. the overall trend shows in the ice extent. to address the trend, scientists need to ask themselves, one direct consequence of the trend is that
Answers: 1


Chemistry, 22.06.2019 18:00, darrell1168
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3

Chemistry, 22.06.2019 23:00, tovarclaudia055
What does a numerical subscript following an element in a chemical formula mean?
Answers: 1
You know the right answer?
Draw a structure for an alcohol that exhibits a molecular ion at m+ = 74 and that produces fragments...
Questions in other subjects:

History, 12.07.2019 10:40

Mathematics, 12.07.2019 10:40


Chemistry, 12.07.2019 10:40

Chemistry, 12.07.2019 10:40

History, 12.07.2019 10:40


Mathematics, 12.07.2019 10:40

History, 12.07.2019 10:40