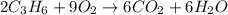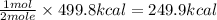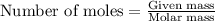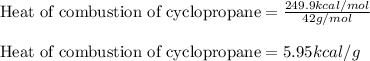Chemistry, 24.01.2020 19:31 sadfaceemojis8300
Combustion reactions are exothermic. the heat of reaction for the combustion of cyclopropane, c3h6, is 499.8 kcal/mol. what is the heat of combustion for cyclopropane in kcal/gram?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, Cooldude3966
As you move from right to left on the periodic table the atomic radius fill in the blank
Answers: 2

Chemistry, 21.06.2019 22:30, kiki197701
Agas at 155 kpa and standard temperature has an initial volume of 1.00 l. the pressure of the gas rises to 500 kpa as the temperature also rises to 135°c. what is the new volume? 2.16 l 0.463 l 0.207 l 4.82 l
Answers: 3

Chemistry, 22.06.2019 00:30, Bryanguzman2004
Water (2510 g ) is heated until it just begins to boil. if the water absorbs 5.09×105 j of heat in the process, what was the initial temperature of the water?
Answers: 3
You know the right answer?
Combustion reactions are exothermic. the heat of reaction for the combustion of cyclopropane, c3h6,...
Questions in other subjects:


Mathematics, 30.09.2019 04:00


History, 30.09.2019 04:00

Mathematics, 30.09.2019 04:00



History, 30.09.2019 04:00


Mathematics, 30.09.2019 04:00