Fe(s) + 2hcl(aq) --> fecl2(aq) + h2(g)
when a student adds 30.0 ml of 1.00 m hcl to...

Chemistry, 23.01.2020 03:31 coollid876
Fe(s) + 2hcl(aq) --> fecl2(aq) + h2(g)
when a student adds 30.0 ml of 1.00 m hcl to 0.56 g of powdered fe, a reaction occurs according to the equation above. when the reaction is complete at 273 k and 1.0 atm, which of the following is true?
a) hcl is in excess, and 0.100 mol of hcl remains unreacted.
d) 0.22 l of h2 has been produced.
the correct answer is d. i can't figure out why a is wrong.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:30, Priskittles
What state of matter is ice a. liquid b. element c. solid d. gas
Answers: 1


Chemistry, 23.06.2019 06:20, ratpizza
Examine the false statement. compounds are the smallest unit of an element that occur most commonly in nature. select the rewording of the statement that is true. a: atoms are the smallest unit of an element that commonly occur in nature. b: molecules are the smallest unit of an element or compound that commonly occur in nature. c: molecules are the smallest unit of a compound that occur on the periodic table. d: compounds are the smallest unit of an element that occur on the periodic table
Answers: 1
You know the right answer?
Questions in other subjects:





History, 28.04.2021 08:00

English, 28.04.2021 08:00


Arts, 28.04.2021 08:00


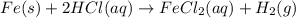
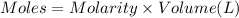
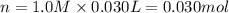
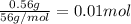
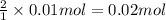 of HCl
of HCl

