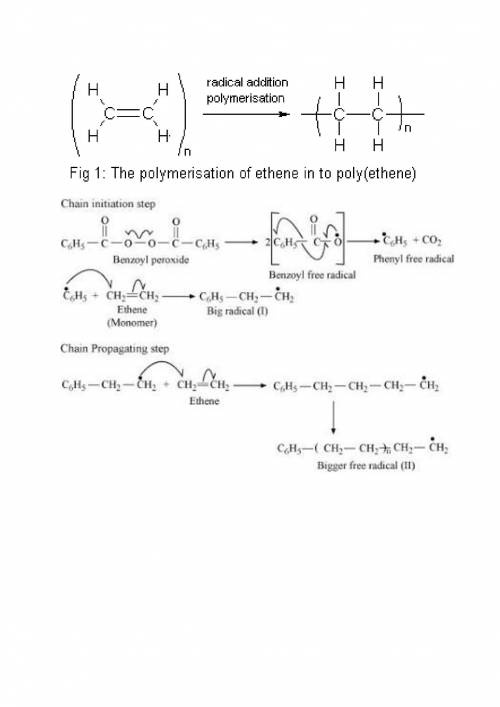
Chemistry, 20.01.2020 20:31 johnnyboy41706
The first step in the free radical mechanism for the preparation of polyethylene is:
a. a formation of a stable carbocation
b. heating an organic peroxide to break the o-o bond
c. formation of an stable carbanion
d. propagation of the free radicals

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 19:00, innocentman69
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1

Chemistry, 22.06.2019 21:00, lucyamine0
As we move from left to right across the periodic table, what is the general trend? a) atomic radii increase. b) electronegavitiy decreases. c) nuclear shielding increases. d) metallic character decreases.
Answers: 1
You know the right answer?
The first step in the free radical mechanism for the preparation of polyethylene is:
a. a for...
a. a for...
Questions in other subjects:



English, 28.06.2019 08:20


English, 28.06.2019 08:20