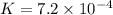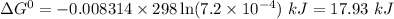Answers: 3


Other questions on the subject: Chemistry



Chemistry, 23.06.2019 04:20, tyrickdavis1
The equation below shows a chemical reaction. a + b + heat —> c + d according to the law of conservation of energy, which statement is true? a. the reactants absorb heat because they have less energy than the products. b. the products release heat because they have more energy than the reactants. c. the reactants generate heat because they have more energy than the products. d. the products require heat to form because they have less energy than the reactants.
Answers: 1
You know the right answer?
Calculate δg° for the following reaction from the equilibrium constant at the temperature given. hf(...
Questions in other subjects:

Mathematics, 20.05.2021 08:20


Engineering, 20.05.2021 08:20

English, 20.05.2021 08:20


Mathematics, 20.05.2021 08:20

Computers and Technology, 20.05.2021 08:20

Mathematics, 20.05.2021 08:20

History, 20.05.2021 08:20

Mathematics, 20.05.2021 08:20

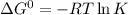
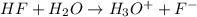
![25^oC=[25+273]K=298K](/tpl/images/0462/9739/df1f6.png)