The missing reagents are.
Potassium + Iodine = Potassium iodide
Calcium + oxygen = Calcium oxide
Beryllium + Bromine = Beryllium bromide
Copper + Oxygen = Copper oxide
Explanation:
The balanced equation can be written as:
1.Potassium + Iodine = Potassium iodide
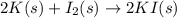
Here K = potassium
I2 = Iodine
KI = potasssium iodide.
2.Calcium + oxygen = Calcium oxide
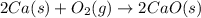
Ca = calcium
O2 = oxygen
CaO = Calcium Oxide
3.Beryllium + Bromine = Beryllium bromide
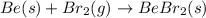
Here,
Be = beryllium
Br2 = bromine
BeBr2 = Beryllium Bromide
4. Copper + Oxygen = Copper oxide
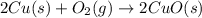
Cu = Copper
O2 = oxygen
CuO = Copper Oxide

















