Chemistry, 15.01.2020 00:31 alexis9263
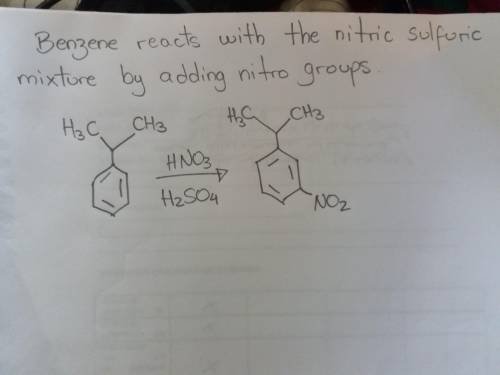
Cumene (isopropylbenzene) is a relatively inexpensive commercially available starting material. show how you could prepare m-isopropylnitrobenzene from cumene.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:00, familyvazquez7
What is the temperature of 0.750 mol of a gas stored in a 6,850 ml cylinder at 2.21 atm? . 2.95 k 5.24 k 138 k 246 k
Answers: 3

Chemistry, 23.06.2019 03:00, draveon6925
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2
You know the right answer?
Cumene (isopropylbenzene) is a relatively inexpensive commercially available starting material. show...
Questions in other subjects:



History, 19.07.2019 20:40


Mathematics, 19.07.2019 20:40




English, 19.07.2019 20:40

Health, 19.07.2019 20:40