
In an experiment, potassium chlorate decomposed according to the following chemical equation. kclo3 → kcl + o2 (molar mass of kclo3 = 122.5 g/mol; kcl = 74.55 g/mol; o2 = 31.998 g/mol) if the mass of kcl produced was 180 grams, which of the following calculations can be used to determine the mass of potassium chlorate decomposed? (180 × 2 × 74.55) ÷ (122.5 × 2) grams (180 × 3 × 74.55) ÷ (122.5 × 2) grams (180 × 2 × 122.5) ÷ (74.55 × 2) grams (180 × 3 × 122.5) ÷ (74.55 × 2) grams

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, Pizzapegasus1
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 10:30, tjjjjjjjjjjjjjjjjjjj
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1

You know the right answer?
In an experiment, potassium chlorate decomposed according to the following chemical equation. kclo3...
Questions in other subjects:



Mathematics, 15.06.2021 01:00


Mathematics, 15.06.2021 01:00


Mathematics, 15.06.2021 01:00

Physics, 15.06.2021 01:00

English, 15.06.2021 01:00

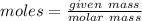
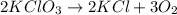
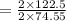 of KClO3
of KClO3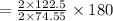 of KClO3
of KClO3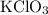 has been given by
has been given by 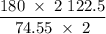 grams. Thus option C is correct.
grams. Thus option C is correct.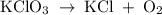
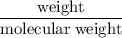
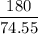 moles
moles molecular weight
molecular weight

