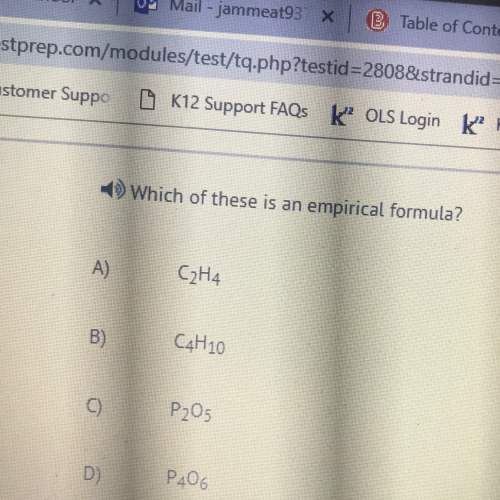
Chemistry, 06.10.2019 21:00 kendallnowell18
Substance x has a fixed volume, and the attraction between its particles is strong. substance y has widely spread out particles and can be compressed. what can most likely be concluded about these substances?
a) substance x is a crystal, and substance y is a liquid.
b) substance x is a gas, and substance y is a plasma.
c) substance x is a plasma, and substance y is a crystal.
d) substance x is a solid, and substance y is a plasma.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, shreyapatel2004
How many atoms of oxygen are contained in 160 grams of n2o3
Answers: 2

Chemistry, 21.06.2019 23:30, hellokitty1647
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i. e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3

Chemistry, 22.06.2019 03:30, electrofy456
What diagram shows the ionic compound of magnesium oxide
Answers: 2

Chemistry, 22.06.2019 05:40, Maryjasmine8001
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2
You know the right answer?
Substance x has a fixed volume, and the attraction between its particles is strong. substance y has...
Questions in other subjects:



Engineering, 18.11.2020 19:30

History, 18.11.2020 19:30




Mathematics, 18.11.2020 19:30

Mathematics, 18.11.2020 19:30

Mathematics, 18.11.2020 19:30