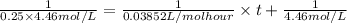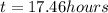At 25 deg. c, the second-order reaction nocl(g)no(g)+
1/2 cl2(g) is 50% complete after 5.82 ho...

Chemistry, 09.01.2020 07:31 genyjoannerubiera
At 25 deg. c, the second-order reaction nocl(g)no(g)+
1/2 cl2(g) is 50% complete after 5.82 hours when the
initialconcentration of nocl is 4.46 mol/l. how long will it
takefor the reaction to be 75% complete?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, Dkhaurithompson
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 08:30, ayaanwaseem
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 10:00, 2019reynolds
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1

Chemistry, 22.06.2019 10:20, blondielocks2002
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3
You know the right answer?
Questions in other subjects:


Mathematics, 06.11.2020 20:30



Mathematics, 06.11.2020 20:30



Social Studies, 06.11.2020 20:30


Chemistry, 06.11.2020 20:30

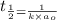
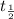 = half life = 5.82 hour
= half life = 5.82 hour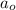 = initial concentration = 4.46 mol/L
= initial concentration = 4.46 mol/L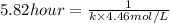
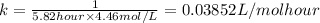
![\frac{1}{[a]}=kt+\frac{1}{[a_o]}](/tpl/images/0448/2354/dfc33.png)
![100\%-75\%=25\% of [a_o]=0.25[a_o]](/tpl/images/0448/2354/5eba6.png)