1) given the balance equation below. calculate how much na3po4 in grams you
would need to creat...

Chemistry, 07.01.2020 19:31 dyanaycooper13
1) given the balance equation below. calculate how much na3po4 in grams you
would need to create 100 grams of naoh.
1 na3po4 + 3 koh → 3 naoh + 1 k3po4

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:00, taylorpayne525p8qxky
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2

Chemistry, 23.06.2019 13:30, leianagaming
1. what is boyle’s law? • state the definition of the law in words. • what are the assumptions of boyle’s law? • write at least one mathematical equation that represents the law. • what can be calculated with boyle’s law? • using a gas-filled balloon as an example, describe what is happening to the gas molecules inside the balloon before and after you squeeze it.
Answers: 2

Chemistry, 23.06.2019 15:30, cicilee49
In a modern periodic table, there are seven periods. a period is any horizontal row of the periodic table, and the elements in a period have consecutive atomic numbers. a group is any vertical column in the periodic table, and there are 18 such groups. groups 3–12, also known as the “b” group elements, are called transition metals. groups 1–2 and 13–18, also known as the “a” group elements, are sometimes called the main groups. metals are characterized by malleability, ductility, conductivity, and a tendency to lose electrons. main group metals are found in groups 1 and 2. nonmetallic elements fall on the right-hand side of the periodic table, that is, groups 13–18. nonmetals have the tendency to gain electrons and are generally brittle. they can be solids, liquids, or gasses at room temperature. now, label the areas of the modern periodic table using the above information. drag the appropriate labels to their respective targets.
Answers: 1
You know the right answer?
Questions in other subjects:



History, 20.09.2020 01:01




Mathematics, 20.09.2020 01:01

Social Studies, 20.09.2020 01:01


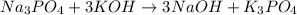
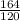
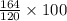 gram of Na3PO4
gram of Na3PO4


