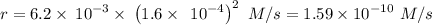The second-order rate constant for the dimerization of a protein (p) p + p → p2 is 6.2 × 10−3/m · s at 25°c. part 1 out of 2 if the concentration of the protein is 1.6 × 10−4 m, calculate the initial rate (m/s) of formation of p2. rate = × 10 m/s (enter your answer in scientific notation.)

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:20, Brooke7644
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 03:00, Dkhaurithompson
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

You know the right answer?
The second-order rate constant for the dimerization of a protein (p) p + p → p2 is 6.2 × 10−3/m · s...
Questions in other subjects:


English, 06.03.2020 23:01





Mathematics, 06.03.2020 23:01

History, 06.03.2020 23:01


Mathematics, 06.03.2020 23:01

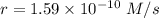
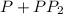
![r=k[P]^2](/tpl/images/0428/7363/45884.png)
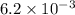 /Ms
/Ms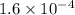 M
M