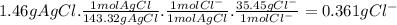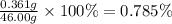To measure the amount of chlorine in a well-boring fluid, an analytical chemist adds 0.1600 m silver nitrate (agno3) solution to a 46.00 g sample of the fluid and collects the solid silver chloride (agcl) product. when no more agcl is produced, he filters, washes and weighs it, and finds that 1.46 g has been produced.
the balanced chemical equation for the reaction is:
cl^- (aq) + agno3(aq) > agcl(s) + no3^- (aq)
1. what kind of reaction is this?
o precipitation o acid-base o redox
2. if you said this was a precipitation reaction, enter the chemical formula of the precipitate.
3. if you said this was an acid-base reaction, enter the chemical formula of the reactant that is acting as the base.
4. if you said this was a redox reaction, enter the chemical symbol of the element that is oxidized.
5. calculate the mass percent of cl in the sample. be sure your answer has the correct number of significant digits.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:20, pandaman632
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1


Chemistry, 22.06.2019 19:00, andrecoral105
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1

Chemistry, 23.06.2019 01:00, stefaniethibodeaux
Substance 33°f 100°f peanut oil solid liquid margarine solid liquid chocolate chips solid liquid which conclusion fits the data in the table? a. heat chemically changes chocolate and margarine. b. all solids become liquid at 100°f. c. removing heat from a substance it to melt. d. matter may change shape when it is heated.
Answers: 1
You know the right answer?
To measure the amount of chlorine in a well-boring fluid, an analytical chemist adds 0.1600 m silver...
Questions in other subjects:

Chemistry, 28.10.2019 04:31


Mathematics, 28.10.2019 04:31



Mathematics, 28.10.2019 04:31

Mathematics, 28.10.2019 04:31

Biology, 28.10.2019 04:31


Chemistry, 28.10.2019 04:31