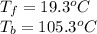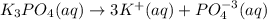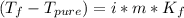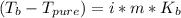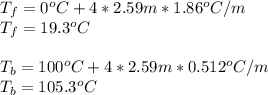Chemistry, 26.10.2019 21:43 therealnana
Assuming 100% dissociation, calculate the freezing point and boiling point of 2.59 m k3po4(aq).

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, penny3109
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 05:30, jzjajsbdb8035
Which other elements contain the same number of outer electrons as sodium
Answers: 3


Chemistry, 22.06.2019 08:00, flakko1899
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1
You know the right answer?
Assuming 100% dissociation, calculate the freezing point and boiling point of 2.59 m k3po4(aq)....
Questions in other subjects:

History, 06.04.2021 17:20


Mathematics, 06.04.2021 17:20

Computers and Technology, 06.04.2021 17:20





Biology, 06.04.2021 17:20

Mathematics, 06.04.2021 17:20