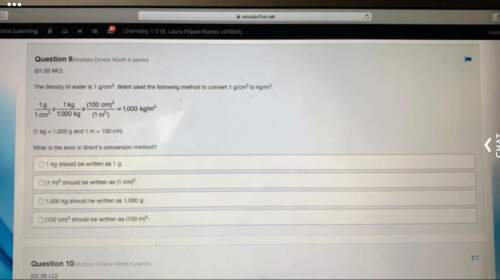
Chemistry, 19.12.2019 01:31 twrxght6643
For the elementary process n2o5(g)−→−no2(g)+no3(g) the activation energy (ea) and overall δe are 154kj/mol and 136kj/mol, respectively. (a) sketch the energy profile for this reaction, and label ea and δe. (b) what is the activation energy for the reverse reaction?

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:30, kittybatch345
Is a chemical message sent by another individual.
Answers: 1


You know the right answer?
For the elementary process n2o5(g)−→−no2(g)+no3(g) the activation energy (ea) and overall δe are 154...
Questions in other subjects:

Business, 08.07.2019 17:50

History, 08.07.2019 17:50




History, 08.07.2019 17:50

Mathematics, 08.07.2019 17:50

History, 08.07.2019 17:50

History, 08.07.2019 17:50