Chemistry, 14.12.2019 07:31 live4dramaoy0yf9
Predicting nuclear stability is important when determining whether or not a nuclear reaction can take place spontaneously. use the following set of guidelines to predict the nuclear stability of the elements listed below.
1. stable nuclei have a high neutron-to-proton ratio, greater than 1.25 beyond atomic number 40 and continuing to rise at higher atomic numbers.
2. the majority of stable nuclei have even numbers of neutrons and protons. odd-even and even-odd combinations can be stable but stability is less likely.
3. nuclei that have the magic numbers of 2, 8, 20, 28, 50, or 82 protons or 2, 8, 20, 28, 50, 82 or 126 neutrons are more stable than nuclei that do not contain these numbers.
4. nuclei that have atomic numbers greater than 83 are unstable. nuclei are considered stable if one or more criteria are met.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:30, nekathadon
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 22.06.2019 17:10, mikeeway33
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

Chemistry, 22.06.2019 18:10, bri9263
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3
You know the right answer?
Predicting nuclear stability is important when determining whether or not a nuclear reaction can tak...
Questions in other subjects:

English, 06.07.2019 13:30





Mathematics, 06.07.2019 13:30


Arts, 06.07.2019 13:30


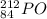
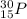
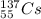
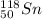
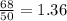 .It is more than 1.25.
.It is more than 1.25.