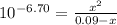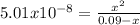* Before addition of any KOH:
pH = 0,0301
*After addition of 25.0 mL KOH:
pH = 1,30
*After addition of 50.0 mL KOH:
pH = 2,87
*After addition of 75.0 mL KOH:
pH = 6,70
*After addition of 100.0 mL KOH:
pH = 10,7
Explanation:
H₃PO₃ has the following equilibriums:
H₃PO₃ ⇄ H₂PO₃⁻ H⁺
k = [H₂PO₃⁻] [H⁺] / [H₃PO₃] k = 10^-(1,30) (1)
H₂PO₃⁻ ⇄ HPO₃²⁻ + H⁺
k = [HPO₃²⁻] [H⁺] / [H₂PO₃⁻] k = 10^-(6,70) (2)
Moles of H₃PO₃ are:
0,0500L×(1,8mol/L) = 0,09 moles of H₃PO₃
* Before addition of any KOH:
Using (1), moles in equilibrium are:
H₃PO₃: 0,09-x
H₂PO₃⁻: x
H⁺: x
Replacing:

4.51x10⁻³ - 0.050x -x² = 0
The right solution of x is:
x = 0.0466589
As volume is 0,050L
[H⁺] = 0.0466589moles / 0,050L = 0,933M
As pH = -log [H⁺]
pH = 0,0301
*After addition of 25.0 mL KOH:
0,025L×1,8M = 0,045 moles of KOH that reacts with H₃PO₃ thus:
KOH + H₃PO₃ → H₂PO₃⁻ + H₂O
That means moles of KOH will be the same of H₂PO₃⁻ and moles of H₃PO₃ are 0,09moles - 0,045moles = 0,045moles
Henderson-Hasselbalch formula is:
pH = pka + log₁₀ [A⁻] /[HA]
Where A⁻ is H₂PO₃⁻ and HA is H₃PO₃.
Replacing:
pH = 1,30 + log₁₀ [0,045mol] / [0,045mol]
pH = 1,30
*After addition of 50.0 mL KOH:
The addition of 50.0 mL KOH consume all H₃PO₃. Thus, in the solution you will have just H₂PO₃⁻. Thus, moles in solution for the equilibrium will be:
H₂PO₃⁻: 0,09-x
HPO₃²⁻: x
H⁺: x
Replacing:
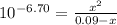
1.8x10⁻⁸ - 2x10⁻⁷x - x² = 0
The right solution of x is:
x = 0.000134064
As volume is 50,0mL + 50,0mL = 100,0mL
[H⁺] = 0.000134064moles / 0,100L = 1.34x10⁻³M
As pH = -log [H⁺]
pH = 2,87
*After addition of 75.0 mL KOH:
Applying Henderson-Hasselbalch formula you will have 0,045 moles of both H₂PO₃⁻ HPO₃²⁻ and pka: 6,70:
pH = 6,70 + log₁₀ [0,045mol] / [0,045mol]
pH = 6,70
*After addition of 100.0 mL KOH:
You will have just 0,09moles of HPO₃²⁻, the equilibrium will be:
HPO₃²⁻ + H₂O ⇄ H₂PO₃⁻ + OH⁻ with kb = kw/ka = 1x10⁻¹⁴/10^-(6,70) = 5,01x10⁻⁸
kb = [H₂PO₃⁻] [OH⁻] / [HPO₃²⁻]
Moles are:
H₂PO₃⁻: x
OH⁻: x
HPO₃²⁻: 0,09-x
Replacing:
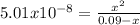
4.5x10⁻⁹ - 5.01x10⁻⁸x - x² = 0
The right solution of x is:
x = 0.000067057
As volume is 50,0mL + 100,0mL = 150,0mL
[OH⁻] = 0.000067057moles / 0,150L = 4.47x10⁻⁴M
As pH = 14-pOH; pOH = -log [OH⁻]
pH = 10,7
I hope it helps!