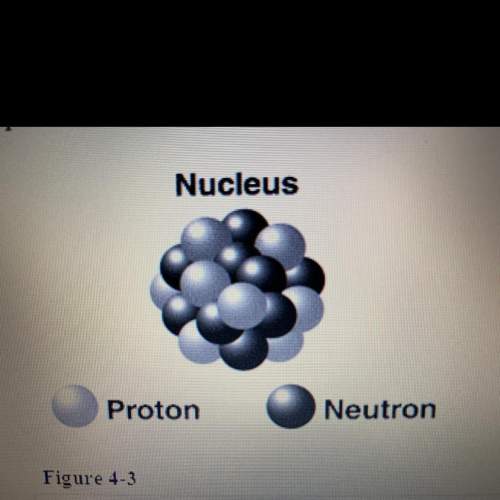
What is the charge on the subatomic particles represented in figure 4-3? assuming all the
par...

Chemistry, 12.12.2019 22:31 shacarabrown6480
What is the charge on the subatomic particles represented in figure 4-3? assuming all the
particles in the nucleus are visible, what are the atomic and mass numbers of the atom shown?
(4 points) **extended response ? **

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:00, anonymous176
Iknow the answer to 13 is b and 14 is d. i just need to know why the correct answers are correct
Answers: 3

Chemistry, 22.06.2019 12:00, hannah2757
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1


Chemistry, 22.06.2019 23:00, lufung8627
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1
You know the right answer?
Questions in other subjects:

Health, 18.07.2019 00:50



Mathematics, 18.07.2019 00:50



Biology, 18.07.2019 00:50



English, 18.07.2019 00:50



