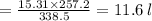Chemistry, 12.12.2019 05:31 laykaleb086
Asample of ammonia gas at 65.5°c and 524 torr has a volume of 15.31 l. what is its volume when the temperature is –15.8°c and its pressure is 524 torr? 20.2 l 11.6 l 63.5 l not possible, since the volume would have to be negative 3.69 l

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:50, Pookiev
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1

Chemistry, 22.06.2019 22:30, wpatskiteh7203
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1

Chemistry, 22.06.2019 23:10, carmenguabaoql9kv
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium. b)heavier than helium. c)the same weight as helium. d)dependent on the element that reacted with carbon.
Answers: 3
You know the right answer?
Asample of ammonia gas at 65.5°c and 524 torr has a volume of 15.31 l. what is its volume when the t...
Questions in other subjects:



Mathematics, 10.12.2020 20:30

Mathematics, 10.12.2020 20:30

Mathematics, 10.12.2020 20:30

History, 10.12.2020 20:30

Arts, 10.12.2020 20:30

Social Studies, 10.12.2020 20:30


Chemistry, 10.12.2020 20:30