Chemistry, 12.12.2019 04:31 kenziepickup
19
select the correct answer from each drop-down menu.
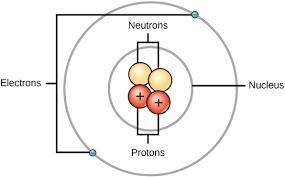
, and the outer part of the
much of the mass of an atom is concentrated in its nucleus. the nucleus contains
atom contains
neutrons and electrons
protons and electrons
protons and neutrons

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 17:00, alevans7144
Why do sodium and neon have vastly different chemical and physical properties despite having similar atomic masses?
Answers: 2

Chemistry, 22.06.2019 03:40, allyyzz
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3

Chemistry, 22.06.2019 09:40, gonzaleze18
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2
You know the right answer?
19
select the correct answer from each drop-down menu.
, and the outer part of the
...
select the correct answer from each drop-down menu.
, and the outer part of the
...
Questions in other subjects:


French, 06.06.2020 03:00

History, 06.06.2020 03:00

Chemistry, 06.06.2020 03:00

Mathematics, 06.06.2020 03:00

Mathematics, 06.06.2020 03:00



Mathematics, 06.06.2020 03:00

English, 06.06.2020 03:00