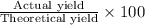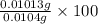Chemistry, 11.12.2019 04:31 jessicalivchits8486
Avolume of 129 ml of hydrogen is collected over water. the water level in the collecting vessel is the same as the outside level. atmospheric pressure is 756.0 torr and the temperature is 25 ∘ c. what is the percent yield of hydrogen for this reaction?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, ruleolivas
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 12:00, 1963038660
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1

Chemistry, 22.06.2019 16:50, lilblackbird4
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3
You know the right answer?
Avolume of 129 ml of hydrogen is collected over water. the water level in the collecting vessel is t...
Questions in other subjects:

Mathematics, 17.03.2021 23:50

Mathematics, 17.03.2021 23:50

Mathematics, 17.03.2021 23:50

Computers and Technology, 17.03.2021 23:50

Chemistry, 17.03.2021 23:50

English, 17.03.2021 23:50


Mathematics, 17.03.2021 23:50

Mathematics, 17.03.2021 23:50

Health, 17.03.2021 23:50

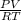
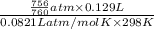
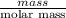
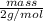
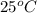
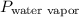 = 24 mm Hg
= 24 mm Hg atm
atm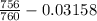
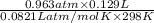
 = 0.0056 mol × 2
= 0.0056 mol × 2