
Chemistry, 11.12.2019 02:31 samueltaye
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.
co(g) + h2o(g) < => co2(g) + h2(g)(volume is decreased)pcl3(g) + cl2(g) < => pcl5(g)(volume is increased)caco3(s)< => cao(s) + co2(g)(volume is increased)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:30, ashtonbillups
Is this a scientific model? use complete sentences to explain why or why not. a graphic organizer showing the water cycle
Answers: 3

Chemistry, 22.06.2019 08:00, juliannxkim
Joe shines white light into a bowl half full of water at an angle of incident of 27.5°. calculate the angle of refraction in the water given the indices of refraction for air and water are 1.00 and 1.36, respectively.
Answers: 2

You know the right answer?
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as...
Questions in other subjects:

History, 16.04.2020 04:45






Mathematics, 16.04.2020 04:45



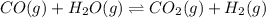 (volume is decreased)
(volume is decreased)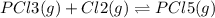 (volume is increased)
(volume is increased)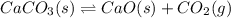 (volume is increased)
(volume is increased)

