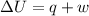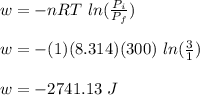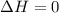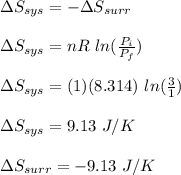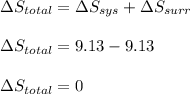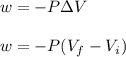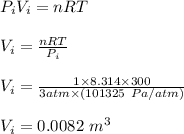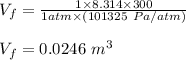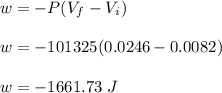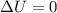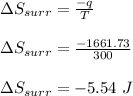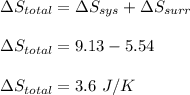Asample consisting of 1.00 mol of perfect gas molecules at 27 °c is expanded isothermally from an initial pressure of 3.00 atm to a final pressure of 1.00 atm in two ways: (a) reversibly, and (b) against a constant external pressure of 1.00 atm. evaluate q, w, δu, δh, δs, δssurr, and δstot in each case.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, mandy9386
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1



Chemistry, 23.06.2019 01:30, bgarrison364
What is produced from neutralization of an acid and a base? a. hydronium ions b. citric acid c. salt and water
Answers: 1
You know the right answer?
Asample consisting of 1.00 mol of perfect gas molecules at 27 °c is expanded isothermally from an in...
Questions in other subjects:



Mathematics, 06.10.2020 15:01

Biology, 06.10.2020 15:01

Biology, 06.10.2020 15:01

History, 06.10.2020 15:01

English, 06.10.2020 15:01


Mathematics, 06.10.2020 15:01

English, 06.10.2020 15:01

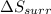 is -9.13 J/K, the entropy change of the system,
is -9.13 J/K, the entropy change of the system, 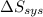 is 9.13 J/K, and the total entropy change,
is 9.13 J/K, and the total entropy change, 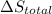 is 0.
is 0.