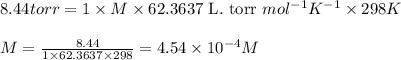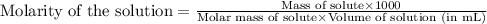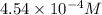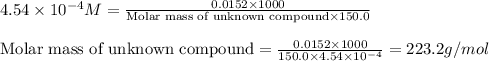Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, bobbycisar1205
How does a hydroelectric power plant converts energy into energy.
Answers: 1

Chemistry, 22.06.2019 14:30, Dreynolds1667
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1

Chemistry, 23.06.2019 10:20, Thejollyhellhound20
Based on the equation, how many grams of br2 are required to react completely with 29.2 grams of alcl3? alcl3 + br2 → albr3 + cl2 48.7 grams 52.6 grams 56.7 grams 61.3 grams
Answers: 3

Chemistry, 23.06.2019 11:50, halllawson
What is the oxidation half-reaction for this unbalanced redox equation? cr2o72– + fe2+ → cr3+ + fe3+ cr3+ → cr2o72– cr2o72– → cr3+ fe3+ → fe2+ fe2+ → fe3+?
Answers: 2
You know the right answer?
A150.0 ml sample of an aqueous solution at 25°c contains 15.2 mg of an unknown nonelectrolyte compou...
Questions in other subjects:



Mathematics, 28.01.2021 01:00

Mathematics, 28.01.2021 01:00

Geography, 28.01.2021 01:00

Mathematics, 28.01.2021 01:00

Mathematics, 28.01.2021 01:00



History, 28.01.2021 01:00

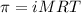
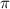 = osmotic pressure of the solution = 8.44 torr
= osmotic pressure of the solution = 8.44 torr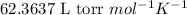
![25^oC=[273+25]=298K](/tpl/images/0411/3120/6a9f9.png)