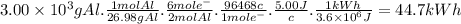The most useful ore of aluminum is bauxite, in which al is present as hydrated oxides, al2o3 * xh2o.
a) the number of kilowatt-hours of electricity required to produce 3.00kg of aluminum from electrolysis of compounds from bauxite is when the applied emf is 5.00 v.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:30, Schoolworkspace453
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2



You know the right answer?
The most useful ore of aluminum is bauxite, in which al is present as hydrated oxides, al2o3 * xh2o....
Questions in other subjects:

Mathematics, 09.10.2020 07:01





Physics, 09.10.2020 07:01

Mathematics, 09.10.2020 07:01