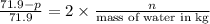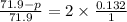Asolution of sodium chloride in water has a vapor pressure of 17.5 torr at 25°c. what is the mole fraction of nacl solute particles in this solution? what would be the vapor pressure of this solution at 45°c? the vapor pressure of pure water is 23.8 torr at 25°c and 71.9 torr at 45°c and assume sodium chloride exists as na⁺ and cl⁻ ions in solution.

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 18:00, heggestade
To apply in a gold the individual gold atoms are united to each other by means of a metallic bond. how would you use the gold block to determine the atomic radius of a gold atom?
Answers: 3
You know the right answer?
Asolution of sodium chloride in water has a vapor pressure of 17.5 torr at 25°c. what is the mole fr...
Questions in other subjects:

Biology, 11.09.2019 00:30



Biology, 11.09.2019 00:30

History, 11.09.2019 00:30



Computers and Technology, 11.09.2019 00:30


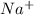 and
and 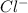 in solution. Therefore, Van't Hoff factor (i) will be equal to 2.
in solution. Therefore, Van't Hoff factor (i) will be equal to 2.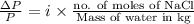
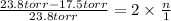
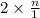
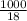
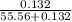
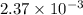
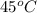 , the vapor pressure will be calculated as follows.
, the vapor pressure will be calculated as follows.