Chemistry, 09.12.2019 20:31 carter4026
The temperature of 2.0 mol of a monatomic ideal gas is 340 k. the internal energy of this gas is doubled by the addition of heat. (a) how much heat is needed when it is added at constant volume? j (b) how much heat is needed when it is added at constant pressure?

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:00, Cythina2007
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

You know the right answer?
The temperature of 2.0 mol of a monatomic ideal gas is 340 k. the internal energy of this gas is dou...
Questions in other subjects:

Mathematics, 01.04.2021 14:10



Mathematics, 01.04.2021 14:20




Social Studies, 01.04.2021 14:20



 = heat at constant volume = ?
= heat at constant volume = ?  = change in internal energy
= change in internal energy = heat capacity at constant volume for monoatomic gas =
= heat capacity at constant volume for monoatomic gas = 
 = initial temperature = 340 K
= initial temperature = 340 K = final temperature =
= final temperature = 





 = heat at constant pressure = ?
= heat at constant pressure = ?  = change in enthalpy energy
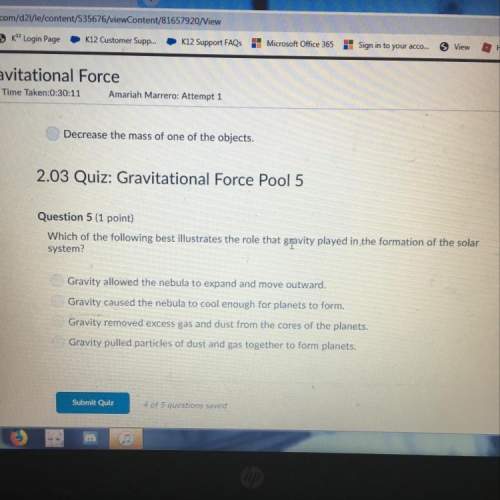
= change in enthalpy energy = heat capacity at constant pressure for monoatomic gas =
= heat capacity at constant pressure for monoatomic gas =