
Chemistry, 09.12.2019 20:31 andrewmena05
What is the rate law for the following mechanism? step1: step2: a+bb+c⇌→cd(fast)(slow) express your answer in terms of k and the necessary concentrations (e. g., k[a]^3[d]).

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, thatonestudent2271
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1



You know the right answer?
What is the rate law for the following mechanism? step1: step2: a+bb+c⇌→cd(fast)(slow) express your...
Questions in other subjects:


Mathematics, 05.11.2020 01:40

Mathematics, 05.11.2020 01:40

Mathematics, 05.11.2020 01:40



History, 05.11.2020 01:40



![Rate=k'[A][B]^2](/tpl/images/0410/3837/02776.png)
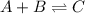 (fast)
(fast)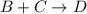 (slow)
(slow)![Rate=k[B][C]](/tpl/images/0410/3837/2d91f.png) ............(1)
............(1)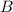 , we get:
, we get:![K=\frac{[C]}{[A][B]}](/tpl/images/0410/3837/f1e20.png)
![[C]=K\times [A][B]](/tpl/images/0410/3837/91a27.png) ...........(2)
...........(2)![Rate=k[B]\times K\times [A][B]](/tpl/images/0410/3837/d3849.png)
![Rate=K\times k[A][B]^2](/tpl/images/0410/3837/56d48.png)



