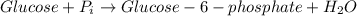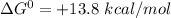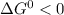Chemistry, 06.12.2019 23:31 deidaralove90
In the first step of glycolysis, the given two reactions are coupled. reaction 1: glucose + p i ⟶ glucose - 6 - phosphate + h 2 o δ g = + 13.8 k j / mol reaction 2: atp + h 2 o ⟶ adp + p i δ g = − 30.5 k j / mol answer the four questions about the first step of glycolysis. is reaction 1 spontaneous or nonspontaneous?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, bibhu42kumarp7o4ss
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1


You know the right answer?
In the first step of glycolysis, the given two reactions are coupled. reaction 1: glucose + p i ⟶ g...
Questions in other subjects:

Mathematics, 26.02.2020 06:01

Mathematics, 26.02.2020 06:01

Mathematics, 26.02.2020 06:01





Mathematics, 26.02.2020 06:02

Biology, 26.02.2020 06:02