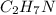Chemistry, 06.12.2019 20:31 heyysiirr3354
Amolecule of ethylamine is made up of two carbon atoms, seven hydrogen atoms, and one nitrogen atom. the molecular formula is c2h7n. the reactive group in ethylamine is nh2. write its condensed structural formula.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, ruleolivas
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 17:30, kiaramccurty
What type of organic molecule comprises the majority of a potato?
Answers: 1

Chemistry, 22.06.2019 19:20, evansh78
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3

Chemistry, 23.06.2019 00:30, lareynademividp0a99r
Gasoline has a density of 0.740 g/ml. if you have 328 grams of gasoline, what is the volume in milliliters?
Answers: 1
You know the right answer?
Amolecule of ethylamine is made up of two carbon atoms, seven hydrogen atoms, and one nitrogen atom....
Questions in other subjects:



Mathematics, 08.12.2021 20:00

Mathematics, 08.12.2021 20:00

History, 08.12.2021 20:00



Mathematics, 08.12.2021 20:00

Social Studies, 08.12.2021 20:00

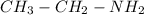 .
.