

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:10, mpchop
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3

Chemistry, 22.06.2019 01:30, lizethdominguez037
What is the value of keq for the reaction expressed in scientific notation
Answers: 1

You know the right answer?
When nh3(g) reacts with o2(g), the products of the combustion are no(g) and h2o(g). what volume of o...
Questions in other subjects:



Mathematics, 29.08.2019 02:50


Health, 29.08.2019 02:50

History, 29.08.2019 02:50

Biology, 29.08.2019 02:50



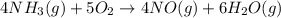
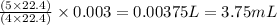 of oxygen gas
of oxygen gas


