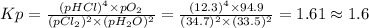Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2(g) + 2h2o(g) → 4hcl(g) + o2(g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound pressure at equilibrium cl2 34.7atm h2o 33.5atm hcl 12.3atm o2 94.9atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant digits.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, Aidanjsauer
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2

Chemistry, 22.06.2019 01:00, angelteddy033
Which of the following is not a true statement about dwarf planets? a the kuiper belt contains comets, asteroids, and dwarf planets. b ceres is a dwarf planet located in the kuiper belt. c the largest known dwarf planet in the solar system is named eris.
Answers: 2

Chemistry, 22.06.2019 07:30, deidaraXneji
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3

Chemistry, 22.06.2019 08:00, anglacx5465
Why are pipes bursting in the in extremely cold weather?
Answers: 2
You know the right answer?
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2(g) + 2h2o(g) → 4hcl(...
Questions in other subjects:

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01