
Silver can be electroplated at the cathode of an electrolysis cell by the half-reaction. ag+(aq)+e−→ag(s) part a silver can be electroplated at the cathode of an electrolysis cell according to the following half-reaction: ag+(aq)+e−→ag(s) what mass of silver plates onto the cathode when a current of 8.5 a flows through the cell for 68 min ?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, actheorian8142
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3

Chemistry, 22.06.2019 12:10, yootmytoot
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution. calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

You know the right answer?
Silver can be electroplated at the cathode of an electrolysis cell by the half-reaction. ag+(aq)+e−→...
Questions in other subjects:




Mathematics, 14.01.2021 21:20

Chemistry, 14.01.2021 21:20

Chemistry, 14.01.2021 21:20

Mathematics, 14.01.2021 21:20



Mathematics, 14.01.2021 21:20

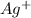 +e− ==> Ag(s)
+e− ==> Ag(s)

