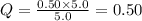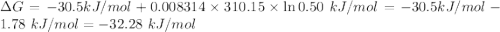Chemistry, 05.12.2019 03:31 chevysilverado3464
For which δg°rxn = –30.5 kj/mol at 37.0 °c and ph 7.0. calculate the value of δgrxn in a biological cell in which [atp] = 5.0 mm, [adp] = 0.50 mm, and [hpo42–] = 5.0 mm.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:40, 19thomasar
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3


Chemistry, 22.06.2019 12:30, americanbellabeauty
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1

Chemistry, 22.06.2019 15:30, sanchez7489
Draw the lewis dot structure for each of the following polyatomic ions
Answers: 1
You know the right answer?
For which δg°rxn = –30.5 kj/mol at 37.0 °c and ph 7.0. calculate the value of δgrxn in a biological...
Questions in other subjects:



Computers and Technology, 10.01.2020 16:31







Arts, 10.01.2020 16:31


 standard Gibbs energy
standard Gibbs energy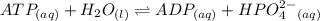
![Q=\frac{[ADP][HPO_4^{2-}]}{[ATP]}](/tpl/images/0403/8875/ccdf0.png)
![[ATP]=5.0 mM](/tpl/images/0403/8875/1ddd2.png)
![[ADP]=0.50 mM](/tpl/images/0403/8875/91d08.png)
![[HPO_4^{2-}]=5.0 mM](/tpl/images/0403/8875/ff97d.png)