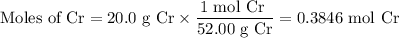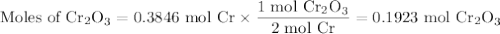Chemistry, 04.12.2019 22:31 KevinReed6444
Given 20.0 of cr, how many grams of the product cr2o3 could be produced?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, boonkgang6821
The clouds are grey and ground is wet. a quantitative b qualitative
Answers: 1


Chemistry, 22.06.2019 22:30, darceline1574
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 22.06.2019 23:00, hailey5campbelp7d1c0
In which region is the substance in both the solid phase and the liquid phase? 1 2. 3 4 mark this and return save and exit next
Answers: 2
You know the right answer?
Given 20.0 of cr, how many grams of the product cr2o3 could be produced?...
Questions in other subjects:

Mathematics, 09.01.2022 08:00



History, 09.01.2022 08:00



Mathematics, 09.01.2022 08:00


History, 09.01.2022 08:00

Mathematics, 09.01.2022 08:00