
Chemistry, 29.11.2019 07:31 tobywaffle1234
Asolution is prepared at 25 °c that is initially 0.23 m in propanoic acid, a weak acid with k, = 1.3 x 10^-5, and 0.42 m in sodium propanoate. calculate the ph of the solution. round your answer to 2 decimal places.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, Bradgarner772
18. use the activity series to predict whether the following synthesis reaction will occur. write the chemical equations for the reaction if it's predicted to occur. (s) + o2(g) -> *note: it is possible.*
Answers: 1

Chemistry, 22.06.2019 06:30, yolo123321
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

You know the right answer?
Asolution is prepared at 25 °c that is initially 0.23 m in propanoic acid, a weak acid with k, = 1.3...
Questions in other subjects:

Chemistry, 11.01.2021 02:10

Mathematics, 11.01.2021 02:10

Chemistry, 11.01.2021 02:10



Mathematics, 11.01.2021 02:10

Mathematics, 11.01.2021 02:10

Mathematics, 11.01.2021 02:10

Mathematics, 11.01.2021 02:10

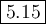

![\begin{array}{rcl}\text{pH} & = & \text{pK}_{\text{a}} + \log \left(\dfrac{[\text{A}^{-}]}{\text{[HA]}}\right )\\\\& = & 4.89 +\log \left(\dfrac{0.42}{0.23}\right )\\\\& = & 4.89 + \log1.83 \\& = & 4.89 +0.261\\& = & 5.15\\\end{array}\\\text{The pH of the buffer is $\large \boxed{\mathbf{5.15}}$}](/tpl/images/0395/8887/e0d2a.png)


