
Chemistry, 28.11.2019 22:31 leannadoughty06
A3.452 g sample containing an unknown amount of a ce(iv) salt is dissolved in 250.0-ml of 1 m h2so4. a 25.00 ml aliquot is analyzed by adding ki and titrating the i3– that forms with s2o32–. the end point was reached following the addition of 13.02 ml of 0.03428 m na2s2o3. calculate the weight percent of ce4 in the sample. when should the indicator be added to this titration? (at the beginning or just before the end point? )

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:10, jasondesatnick
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2


Chemistry, 22.06.2019 17:00, davisnaziyahovz5sk
The arrangement of particles is most ordered in a sample of
Answers: 1
You know the right answer?
A3.452 g sample containing an unknown amount of a ce(iv) salt is dissolved in 250.0-ml of 1 m h2so4....
Questions in other subjects:



Mathematics, 07.05.2021 18:10


Mathematics, 07.05.2021 18:10




Mathematics, 07.05.2021 18:10

Spanish, 07.05.2021 18:10

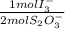 = 2,2315x10⁻⁴ moles of I₃⁻
= 2,2315x10⁻⁴ moles of I₃⁻ = 4,463x10⁻⁴ moles of Ce(IV). These moles are:
= 4,463x10⁻⁴ moles of Ce(IV). These moles are: = 0,0625 g of Ce(IV)
= 0,0625 g of Ce(IV)

