Chemistry, 28.11.2019 07:31 oneicyahdaley10
(1.) using beer's law, how will the absorbance measured for the solutions change as the concentration of aspirin in solutions increase?
(2.) in an experiment, the beer’s law plot resulted in the following relationship between absorbance and concentration of asa, y = 1061.5x, where y is absorbance and x is the concentration. if the absorbance of a sample solution prepared from an aspirin tablet is 0.402, calculate the concentration of asa in the solution in m.
(3.) if the above solution was prepared by taking 10 ml of stock solution and diluting it to 100 ml, what is the concentration of the stock solution?
(4.) this stock solution was prepared as follows: an aspirin tablet was transferred to an erlenmeyer flask and reacted with naoh. the resulting solution was transferred to a 250 ml volumetric flask and the volume made up to 250 ml. calculate the mass of aspirin in the tablet based on the concentration of aspirin in the stock solution.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, nuclearfire278
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease. correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1



Chemistry, 23.06.2019 00:30, quintink
How can you write e method for the experiment of separating sand from water by filtration process? 1-materials 2-steps 3-conclusion also the same for the separating process of water and salt by filtration or distillation. quick because i need to finish my hw
Answers: 2
You know the right answer?
(1.) using beer's law, how will the absorbance measured for the solutions change as the concentratio...
Questions in other subjects:

English, 10.05.2021 22:10

Mathematics, 10.05.2021 22:10

Mathematics, 10.05.2021 22:10

Chemistry, 10.05.2021 22:10

Mathematics, 10.05.2021 22:10

Mathematics, 10.05.2021 22:10



Mathematics, 10.05.2021 22:10

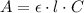 (1)
(1)![A = 1061.5 \cdot [ASA]](/tpl/images/0394/5398/223a2.png)
![[ASA] = \frac{A}{1061.5} = 3.79 \cdot 10^{-4}M](/tpl/images/0394/5398/3c100.png)
![V_{i} [ASA]_{i} = V_{f} [ASA]_{f}](/tpl/images/0394/5398/57531.png)
![[ASA]_{i} = \frac{V_{f} \cdot [ASA]_{f}}{V_{i}} = \frac {100mL \cdot 3.79\cdot 10^{-4} M}{10mL} = 3.79 \cdot 10^{-3} M](/tpl/images/0394/5398/a10be.png)
![m_{ASA} = \eta_{ASA} \cdot M_{ASA} = [ASA]_{i} \cdot V_{0} \cdot M_{ASA}](/tpl/images/0394/5398/9bfc0.png)