
The air bags in automobiles were once inflated by nitrogen gas generated by the rapid decomposition of sodium azide, nan3. if an air bag has a volume of 43.8 l and is to be filled with nitrogen gas at a pressure of 1.13 atm at a temperature of 22.4˚c, how many moles of nan3 must decompose? you may assume the n2 behaves as an ideal gas. if carmen adds zeros behind the decimal place in your answer, do not worry. it will be graded correctly.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, lifeofabe214
Which type of stress results when two plates push against one another? a. compression b. tension c. force d. shear
Answers: 1

Chemistry, 22.06.2019 02:30, Countryqueen525
Asa choose the correct set of reaction coefficients to properly balance the following chemical equation according to the law of conservation of mass: __s8 + __o2 ==> __so2 1, 1, 8 1, 8, 1 1, 8, 8 8, 1, 1
Answers: 1


Chemistry, 22.06.2019 17:00, brandiwingard
What is the mass of phosphorous in a 51-kg person
Answers: 1
You know the right answer?
The air bags in automobiles were once inflated by nitrogen gas generated by the rapid decomposition...
Questions in other subjects:


Social Studies, 28.05.2020 23:59

English, 28.05.2020 23:59



Mathematics, 28.05.2020 23:59

Mathematics, 28.05.2020 23:59

History, 28.05.2020 23:59

Mathematics, 28.05.2020 23:59

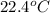 = (22.4 + 273) K = 295.4 K
= (22.4 + 273) K = 295.4 K
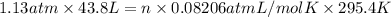
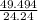
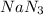 must decompose.
must decompose.

