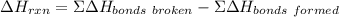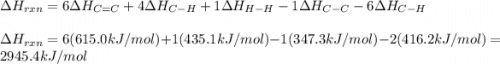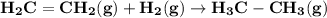Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, kkelley9223
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2


Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1
You know the right answer?
Use average bond energies to calculate δhrxn for the following hydrogenation reaction: h2c=ch2(g)+h...
Questions in other subjects:

Mathematics, 22.01.2021 07:00


Health, 22.01.2021 07:00


Geography, 22.01.2021 07:00

Mathematics, 22.01.2021 07:00




History, 22.01.2021 07:00