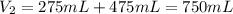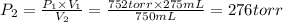A275-ml flask contains pure helium at a pressure of 752 torr. a second flask with a volume of 475 ml contains pure argon at a pressure of 722 torr. if the two flasks are connected through a stopcock and the stopcock is opened, what is the partial pressure of each gas and the total pressure.?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:40, janelisse199820
Non renewable resources like petroleum eventually
Answers: 2

Chemistry, 22.06.2019 02:40, alexandraparava
For a patient with the following pes statement and interventions, which would be the most appropriate monitoring and evaluating data? pes statement: inadequate calcium intake related to food and nutrition related knowledge deficit as evidenced by statements that the only dietary source of calcium is milk and she believes that she is lactose intolerant. patient’s nutrition prescription is for a diet providing 1200 mg calcium per day. patient was provided with in-depth nutrition education on alternative dietary and supplement sources of calcium. a. calcium intake (at subsequent visit) b. knowledge assessment by asking patient to identify food sources from menus and shopping list (at the end of the current visit) c. serum calcium (at next visit) d. both a and b e. both a and c
Answers: 2


Chemistry, 22.06.2019 05:50, vanessa051266
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1
You know the right answer?
A275-ml flask contains pure helium at a pressure of 752 torr. a second flask with a volume of 475 ml...
Questions in other subjects:






Mathematics, 15.07.2021 23:40

History, 15.07.2021 23:40

Chemistry, 15.07.2021 23:40