
Chemistry, 26.11.2019 06:31 jdkrisdaimcc11
The thiocyanate polyatomic ion, scn-, is commanly called a pseudohalogen because it acts very much like halide ions. for example, we know that pure halogens consists of diatiomic molecules, such as c12. thiocanate ions form similar molecules in the following reaction2nascn +2h2so4 + mno2 -> (scn)2 + 2h2o + mnso4 + na2so4a) write a conversion factor that could be used to convert between moles of nascn and moles of (scn)2b) how many moles of (scn)2 form when 0.05 moles of nascn react completely? c) what is the maximum number of moles of (scn)2 that could form in the combination of 4 moles of nascn and 3 moles of mnso4? d) write a convertion factor that could be used to convert between moles of sulfuric acid, h2so4 and moles manganese (ii) sulfate, mnso4.e) what is the minimum number of moles of h2so4 that must react to form 1.7752 moles of manganese (ii) sulfate?

Answers: 3


Other questions on the subject: Chemistry



You know the right answer?
The thiocyanate polyatomic ion, scn-, is commanly called a pseudohalogen because it acts very much l...
Questions in other subjects:






Biology, 04.12.2019 22:31

Mathematics, 04.12.2019 22:31



Chemistry, 04.12.2019 22:31



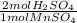 = 3.5504 mol H₂SO₄
= 3.5504 mol H₂SO₄

